2256. Chrysin
Nomenclature
CAS number: 480-40-0
5,7-Dihydroxy-2-phenyl-4H-1-benzopyran-4-one; 5,7-dihydroxyflavone; chrysidenon 1438. C15H10O4; mol wt 254.24.
C 70.86%, H 3.96%, O 25.17%.
Description and references
From heartwood of Pinus monticola Dougl., P. excelsa Wall., and P. aristata Engelm., Pinaceae: Linstedt, Acta Chem. Scand. 3, 1147, 1375 (1949); 4, 55 (1950); from bark of Dolichandrone falcata Seem., Bisnomiaceae: Kincl, Naturwissenschaften 42, 646 (1955). Synthesis: Seka, Prosche, Monatsh. Chem. 69, 284 (1936); Hutchins, Wheeler, J. Chem. Soc. 1939, 91; Teoule et al., Bull. Soc. Chim. Fr. 1961, 546.
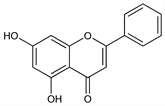
Properties
Light yellow prisms from methanol, mp 285°. uv max: 270, 329 nm (log ε 4.40, 3.90). Practically insol in water; sol in alkali hydroxide solns; slightly sol in alcohol, chloroform, ether.Derivative
Diacetoxychrysin.C19H14O6; mol wt 338.31.
C 67.45%, H 4.17%, O 28.38%.
Properties
Crystals from ethanol, mp 194-195°.Derivative
Methylchrysin.Nomenclature
Tectochrysin.C16H12O4; mol wt 268.26.
C 71.64%, H 4.51%, O 23.86%.
Description and references
It is present as such or in the form of a glucoside in buds of Populus spp., Salicaceae. Use of tectochrysin as diuretic: Perrault, US 3155579 (1964 to Laroche Navarron).