2258. Chrysophanic Acid
Nomenclature
CAS number: 481-74-3
1,8-Dihydroxy-3-methyl-9,10-anthracenedione; 1,8-dihydroxy-3-methylanthraquinone; 3-methylchrysazin; chrysophanol. C15H10O4; mol wt 254.24.
C 70.86%, H 3.96%, O 25.17%.
Description and references
Occurs in the free state and as glucoside in cascara sagrada, senna and various species of Rumex and Rheum (rhubarb). Isoln from rhubarb root: Tutin, Clewer, J. Chem. Soc. 99, 946 (1911); Siesto, Bartoli, Farmaco Ed. Prat. 12, 517 (1957); Carelli, Giuliano, ibid. 184; from Penicillium islandicum Sopp.: Howard, Raistrick, Biochem. J. 46, 49 (1950); from Chaetonium affine Corda: Arkley et al., Croat. Chem. Acta 29, 141 (1957), C.A. 53, 1287h (1959). Synthesis: Eder, Widmer, Helv. Chim. Acta 5, 3 (1922); 6, 419 (1923); Ayyangar et al., J. Sci. Ind. Res. 20B, 493 (1961). Total synthesis: M. E. Jung, J. A. Lowe, Chem. Commun. 1978, 95.
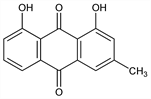
Properties
Hexagonal or monoclinic crystals from alcohol or benzene, mp 196°. Sublimes. Absorption max: 226, 256, 278, 288, 436 nm (ε × 10-3 41, 28, 14, 14, 11.8). Practically insol in water. Slightly sol in cold, freely in boiling alc; sol in benzene, chloroform, ether, glacial acetic acid, acetone, solns of alkali hydrides, and in hot solns of alkali carbonates; very slightly sol in petr ether.Derivative
Glucoside.Nomenclature
Chrysophanein; chrysophaniin.C21H20O9; mol wt 416.38.
C 60.58%, H 4.84%, O 34.58%.