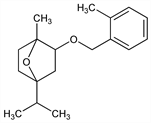
2295. Cinmethylin
Nomenclature
CAS number: 87818-31-3
exo-(±)-1-Methyl-4-(1-methylethyl)-2-[(2-methylphenyl)methoxy]-7-oxabicyclo[2.2.1]heptane; (±)-2-exo-(2-methylbenzyloxy)-1-methyl-4-isopropyl-7-oxabicyclo[2.2.1]heptane; SD-95481; Cinch (DuPont). C18H26O2; mol wt 274.40.
C 78.79%, H 9.55%, O 11.66%.
Description and references
Pre-emergence grass herbicide. Member of the cineole eucalyptol, q.v., family. Prepn: G. B. Payne et al., EP 81893 (1983 to Shell); eidem, US 4670041 (1987 to Du Pont). Physical and chemical properties: B. T. Grayson et al., Pestic. Sci. 21, 143 (1987). Mechanism of action study: M. H. El-Deek, F. D. Hess, Weed Sci. 34, 684 (1986). Metabolism in rats: P. W. Lee et al., J. Agric. Food Chem. 34, 162 (1986). Mobility in soil: D. R. Wendt et al., Proc. South. Weed Sci. Soc. 1987, 391; and herbicidal activity: P. C. Lolas, A. Galopoulos, Zizaniology 1, 221 (1985). Field studies in tobacco: P. C. Lolas, Proc. Br. Crop Prot. Conf. - Weeds 1985, 841; in soybeans: P. C. Bhowmik, Weed Sci. 36, 678 (1988).