2319. Citicoline
Nomenclature
Description and references
Naturally occurring nucleotide; intermediate in the major pathway of lecithin biosynthesis. Identification: E. P. Kennedy, S. B. Weiss, J. Am. Chem. Soc. 77, 250 (1955). Crystallization from yeast extract: I. Lieberman et al., Science 124, 81 (1956). Synthesis: E. P. Kennedy, J. Biol. Chem. 222, 185 (1956); K. Kikugawa et al., Chem. Pharm. Bull. 19, 1011, 2466 (1971). Molecular structure: M. A. Viswamitra et al., Nature 258, 497 (1975). Series of articles on pharmacology and toxicology: Arzneim.-Forsch. 33, 1009-1080 (1983). Acute toxicity: T. Grau et al., ibid. 1033. Clinical trial in ischemic stroke: W. M. Clark et al., Neurology 49, 671 (1997). Review of biosynthesis, metabolism, pharmacology: G. B. Weiss, Life Sci. 56, 637-660 (1995); and clinical experience: J. J. Secades, G. Frontera, Methods Find. Exp. Clin. Pharmacol. 17, Suppl. B, 1-54 (1995).
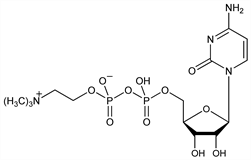
Properties
Amorphous, somewhat hygroscopic powder. [α]D25 +19.0° (c = 0.86 in H2O). uv max (pH 1): 280 nm (ε 12800). Dissolves readily in water to form acidic soln. Practically insol in most organic solvents. pKa 4.4. LD50 in mice, rats (mg/kg): 4600 ±335, 4150 ±370 i.v.; both species 8 g/kg orally (Grau).Derivative
Sodium salt.Nomenclature
Properties
White, crystalline, spongy, hygrosopic powder, dec 250°. [α]D20 +12.5° (c = 1.0 in H2O). Sol in water. Practically insol in alcohol.Therapeutic Category
Neuroprotective. In treatment of ischemic stroke and head trauma.
Keywords
Neuroprotective