2353. Clevudine
Nomenclature
Description and references
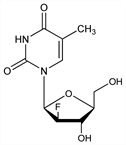
Nucleoside analog; specific inhibitor of viral DNA synthesis. Prepn: C. K. Chu et al., WO 9520595; eidem, US 5587362 (1995, 1996 both to Univ. Georgia Res. Found.; Yale Univ.); T. Ma et al., J. Med. Chem. 39, 2835 (1996). Synthesis: M. L. Sznaidman et al., Nucleosides Nucleotides Nucleic Acids 21, 155 (2002). In vitro antiviral activity: C. K. Chu et al., Antimicrob. Agents Chemother. 39, 979 (1995). Pharmacokinetics: J. D. Wright et al., Pharm. Res. 12, 1350 (1995). Mechanism of action study: Y. Chong, C. K. Chu, Bioorg. Med. Chem. Lett. 12, 3459 (2002). Clinical evaluation in chronic hepatitis B: P. Marcellin et al., Hepatology 40, 140 (2004). Review of preclinical pharmacology: C. K. Chu et al. in Therapies for Viral Hepatitis, R. F. Schinazi et al., Eds. (Int. Med. Press, London, 1998) pp 303-312; of development and clinical experience: G. R. Painter et al. in Frontiers in Viral Hepatitis, R. F. Schinazi et al., Eds. (Elsevier, New York, 2003) pp 281-300.
Properties
Crystals from methanol/chloroform, mp 184-185°. [α]D25 -111.77° (c = 0.23 in methanol). uv max in water: 265.0 (pH 2), 265.5 (pH 7), 265.5 nm (pH 11) (ε 9695, 9647, 7153). LD50 in mice, rats (mg/kg): >5000, >3000 orally (Painter).Derivative
Triphosphate.Nomenclature
Therapeutic Category
Antiviral.
Keywords
Antiviral; Nucleosides/Nucleotides