2389. Clonazepam
Nomenclature
Description and references
Benzodiazepine antiepileptic agent with anxiolytic and antimanic properties. Prepn: L. H. Sternbach et al., J. Med. Chem. 6, 261 (1963); O. Keller et al., US 3121076 (1964 to Hoffmann-La Roche). Prepd not claimed: J. Kariss, H. L. Newmark, US 3116203 (1963 to Hoffmann-La Roche). Alternate process: A. Focella, A. I. Rachlin, US 3335181 (1967 to Hoffmann-La Roche). Pharmacology: Guerrero-Figueroa et al., Curr. Ther. Res. 11, 40 (1969); Lechat et al., Therapie 25, 893 (1970); D'Armagnac et al., ibid. 26, 439 (1971). Toxicology: Blum et al., Arzneim.-Forsch. 23, 377 (1973). HPLC determn in plasma: I. F. Bares et al., J. Pharm. Biomed. Anal. 36, 865 (2004). Comprehensive description: W. C. Winslow, Anal. Profiles Drug Subs. 6, 61-81 (1977). Review of pharmacokinetics: D. J. Greenblatt et al, J. Clin. Psychiatry 48, 4-9 (1987); of clinical efficacy in panic attacks: M. H. Pollack, ibid. 12-14; in acute mania: G. Chouinard, ibid. 29-37. Clinical trial in panic disorders: G. Moroz, J. F. Rosenbaum, ibid. 60, 604 (1999).
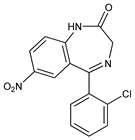
Properties
White crystals from ethanol-methylene chloride, mp 236.5-238.5°. uv max (7.5% methanol in isopropanol): 248, 310 nm (ε 14500, 11600). Soly in mg/ml at 25°: acetone 31; chloroform 15; methanol 8.6; ether 0.7; benzene 0.5; water <0.1. pK1 1.5, pK2 10.5. LD50 orally in mice: >4000 mg/kg (Blum).Note
This is a controlled substance (depressant): 21 CFR, 1308.14.Therapeutic Category
Anticonvulsant.
Therapeutic Category (Veterinary)
Anticonvulsant.Keywords
Anticonvulsant