2417. Clotrimazole
Nomenclature
CAS number: 23593-75-1
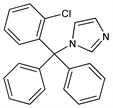
1-[(2-Chlorophenyl)diphenylmethyl]-1H-imidazole; 1-(o-chloro-α,α-diphenylbenzyl)imidazole; 1-[α-(2-chlorophenyl)benzhydryl]imidazole; 1-[(o-chlorophenyl)diphenylmethyl]imidazole; diphenyl-(2-chlorophenyl)-1-imidazolylmethane; 1-(o-chlorotrityl)imidazole; FB-5097; Bay b 5097; Canesten (Bayer); Canifug (Wolff); Empecid (Bayer-Takeda); Gyne-Lotrimin (Schering-Plough); Lotrimin (Schering-Plough); Mono-Baycuten; Mycelex-G (Miles); Mycofug (Hermal); Mycosporin (Bayer); Pedisafe (Sagitta); Rimazole (Cheil Sugar); Tibatin (Dak); Trimysten. C22H17ClN2; mol wt 344.84.
C 76.63%, H 4.97%, Cl 10.28%, N 8.12%.
Description and references
Prepn: K. H. Buechel et al., ZA 6805392; eidem, US 3705172 (1969, 1972 both to Bayer). Pharmacology: Plempel et al., Antimicrob. Agents Chemother. 1969, 271; eidem, Dtsch. Med. Wochenschr. 94, 1356 (1969). Clinical findings: Oberste-Lehn et al., ibid. 1365. Series of articles on prepn, toxicology, pharmacokinetics, clinical studies: Arzneim.-Forsch. 22, 1260-1272, 1276-1299 (1972). Toxicity: D. Tettenborn, ibid. 1276. Comprehensive description: J. G. Hoogerheide, B. E. Wyka, Anal. Profiles Drug Subs. 11, 225-255 (1982).
Properties
Crystals, mp 147-149°. A weak base, slightly sol in water, benzene, toluene; sol in acetone, chloroform, ethyl acetate, DMF. Hydrolyzes rapidly upon heating in aq acids. LD50 in male mice, rats (mg/kg): 923, 708 orally (Tettenborn).Derivative
Hydrochloride.C22H17ClN2.HCl; mol wt 381.30.
C 69.30%, H 4.76%, Cl 18.60%, N 7.35%.
Properties
mp 159°.Therapeutic Category
Antifungal.
Therapeutic Category (Veterinary)
Antifungal.Keywords
Antifungal (Synthetic); Imidazoles