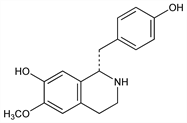
2459. Coclaurine
Nomenclature
CAS number: 486-39-5
(1S)-1,2,3,4-Tetrahydro-1-[(4-hydroxyphenyl)methyl]-6-methoxy-7-isoquinolinol; 1-(p-hydroxybenzyl)-6-methoxy-7-hydroxy-1,2,3,4-tetrahydroisoquinoline; machiline. C17H19NO3; mol wt 285.34.
C 71.56%, H 6.71%, N 4.91%, O 16.82%.
Description and references
Isolated as the racemate from species of Machilus (Lauraceae) and Cocculus (Menispermaceae). First isoln from C. laurifolius D.C. believed to be of the d-form: Kondo, Kondo, J. Pharm. Soc. Jpn. No. 524, 876 (1925), C.A. 20, 6047 (1926); see also Johns et al., Aust. J. Chem. 20, 1729 (1967). Structure: Kondo, Kondo, J. Pharm. Soc. Jpn. 48, 1156 (1928); Tomita, Kusuda, ibid. 72, 280 (1952). Synthesis: Kratzl, Billek, Monatsh. Chem. 82, 568 (1951); Finkelstein, J. Am. Chem. Soc. 73, 550 (1951). Identity with machiline: Tomita et al., J. Pharm. Soc. Jpn. 83, 218 (1963), C.A. 59, 2874a (1963). Crystal structure and absolute configuration: Fridrichsons, Mathieson, Tetrahedron 24, 5785 (1968).
Properties
Plates, tablets from alc, mp 220-221°. Sol in hot alc, hot acetone; slightly sol in water, alc, chloroform, ether, acetone; practically insol in benzene, petr ether.Derivative
Hydrochloride.C17H19NO3.HCl; mol wt 321.80.
C 63.45%, H 6.26%, N 4.35%, O 14.92%, Cl 11.02%.