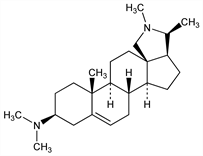
2497. Conessine
Nomenclature
Description and references
Antiamebic, antifungal principle in Kurchi bark, the bark of Holarrhena anti-dysenterica Wall., Apocynaceae, native to India; also found in African species, such as H. africana A.DC.; H. congolensis Stapf; H. wulfsbergii Stapf, and H. febrifuga Klotzsch, Apocynaceae. Isoln: Haines, Trans. Med. Soc. Bombay 4, 28 (1858); Warnecke, Ber. 19, 60 (1886); Bertho, Arch. Pharm. 277, 237 (1939); Siddiqui, Pillay, J. Indian Chem. Soc. 9, 553 (1932). Structure: Haworth et al., J. Chem. Soc. 1953, 1102; Haworth, McKenna, Chem. Ind. (London) 1957, 1510; Bertho, G"otz, Ann. 619, 96 (1958). Discussion of structure in Fieser, Fieser, Steroids (New York, 1959), p 858. Synthesis from Δ5-pregnene-3β,20β-diol: Barton, Morgan, Proc. Chem. Soc. London 1961, 206; J. Chem. Soc. 1962, 622. Total synthesis: Marshall, Johnson, J. Am. Chem. Soc. 84, 1485 (1962); Stork et al., ibid. 2018; Nagata et al., Tetrahedron Lett. 1963, 869.