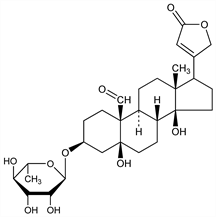
2512. Convallatoxin
Nomenclature
Description and references
From blossoms of lily of the valley (Convallaria majalis L., Liliaceae): Karrer, Helv. Chim. Acta 12, 506 (1929); from Ornithogalum umbellatum L., Liliaceae: Mrozik et al., ibid. 42, 683 (1959); from Antiaris toxicaria Lesch, Moraceae: Juslen et al., ibid. 46, 117 (1963). Structure: Tschesche, Haupt, Ber. 69, 459 (1936); Fieser, Jacobsen, J. Am. Chem. Soc. 59, 2335 (1937). Cleavage into l-rhamnose and strophanthidin: Reichstein, Katz, Pharm. Acta Helv. 18, 521 (1943); see also C.A. 38, 2046. Synthesis from strophanthidin and acetobromrhamnose: Reyle et al., Helv. Chim. Acta 33, 1541 (1950); Haede et al., DE 1933090 (1971 to Hoechst). Toxicity study: W. F"orster et al., Arch. Int. Pharmacodyn. 155, 165 (1965).
Properties
Prisms from methanol + ether. mp 235-242°. [α]D22 -1.7 ± 3° (c = 0.65 in methanol); [α]D25 -9.4 ± 3° (c = 0.72 in dioxane). Sol in alcohol, acetone; slightly sol in chloroform, ethyl acetate, and water (1:2000). Practically insol in ether, petr ether. Gives Legal's reaction. LD50 in mice, rats (mg/kg): 10.0 i.p., 16.0 i.v. (F"orster).Derivative
Tri-O-acetyl-convallatoxin.Properties
Needles from acetone + ether, mp 215-238°. [α]D25 -5.5 ± 2° (c = 0.962 in chloroform).Therapeutic Category
Cardiotonic.
Keywords
Cardiotonic