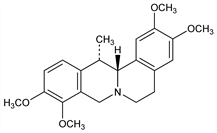
2544. Corydaline
Nomenclature
Description and references
Alkaloid isolated from many species of Corydalis. Discovery in C. tuberosa: Wackenroder, Kustner's Arch. 8, 423 (1826), as cited in: C. Wehmer, Die Pflanzenstoffe I (G. Fischer, Jena, 1929) p 389. Isoln from Corydalis aurea Willd. and C. solida (L.) Swartz, Fumariaceae: Manske, Can. J. Res. 16B, 81 (1938); Can. J. Chem. 34, 1 (1956). Structure: von Bruchhausen, Stippler, Arch. Pharm. 265, 152 (1927). Synthesis: Sp"ath, Kruta, Ber. 62, 1024 (1929); T. Kametani et al., J. Chem. Soc. Perkin Trans. 1 1977, 1151; M. Cushman, F. W. Dekon, Tetrahedron 1978, 1435; Z. Kiparissides et al., Can. J. Chem. 58, 2770 (1980). Stereochemistry: Bersch, Arch. Pharm. 291, 595 (1958); Jeffs, Experientia 21, 690 (1965). Pharmacology: Berezhinskaya, C.A. 78, 119087j (1973). Toxicity study: R. C. Anderson, K. K. Chen, Fed. Proc. 5, 163 (1946).