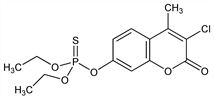
2558. Coumaphos
Nomenclature
CAS number: 56-72-4
Phosphorothioic acid O-(3-chloro-4-methyl-2-oxo-2H-1-benzopyran-7-yl) O,O-diethyl ester; O,O-diethyl O-(3-chloro-4-methyl-2-oxo-2H-1-benzopyran-7-yl)phosphorothioate; O-3-chloro-4-methyl-2-oxo-2H-chromen-7-yl O,O-diethyl phosphorothioate; 3-chloro-7-diethoxyphosphinothioyloxy-4-methylcoumarin; 3-chloro-4-methylumbelliferone O,O-diethyl phosphorothioate; O,O-diethyl O-(3-chloro-4-methyl-7-coumarinyl)
phosphorothioate; coumafos; Bayer 21/199; Asuntol (Bayer); Baymix (Bayer); Co-ral (Bayer); Muscatox (Bayer); Perizin (Bayer); Resitox (BASF). C14H16ClO5PS; mol wt 362.77.
C 46.35%, H 4.45%, Cl 9.77%, O 22.05%, P 8.54%, S 8.84%.
Description and references
Organophosphate insectide; cholinesterase inhibitor. Prepn: Schrader, DE 881194; US 2748146 (1951, 1956 both to Bayer). Metabolism: H. R. Krueger et al., J. Agric. Food Chem. 7, 182 (1959) DOI. GC determn in honey: U. Menkissoglu-Spiroudi et al., J. AOAC Int. 83, 178 (2000) PubMed. Toxicity: T. B. Gaines, Toxicol. Appl. Pharmacol. 14, 515 (1969) DOI PubMed. Efficacy for nematodes in cattle: M. F. Hansen, S. M. Zeakes, Trans. Am. Micros. Soc. 88, 159 (1969) DOI; for cattle ticks: R. B. Davey et al., Prev. Vet. Med. 23, 1 (1995) DOI. Use in eradication of bee mites and distribution in hives: P. Tremolada et al., Ecotoxicology 13, 589 (2004) DOI PubMed.