2564. Coumestrol
Nomenclature
CAS number: 479-13-0
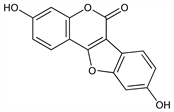
3,9-Dihydroxy-6H-benzofuro[3,2-c][1]benzopyran-6-one; 2-(2,4-dihydroxyphenyl)-6-hydroxy-3-benzofurancarboxylic acid δ-lactone; 7′,6-dihydroxycoumarino(3′,4′,3,2)coumarone. C15H8O5; mol wt 268.22.
C 67.17%, H 3.01%, O 29.83%.
Description and references
An estrogenic factor occurring naturally in forage crops, esp in ladino clover (Trifolium repens L.), strawberry clover (T. fragiferum L.) and alfalfa (Medicago sativa L., Leguminosae). Isoln: Bickoff et al., J. Agric. Food Chem. 6, 536 (1958); Bickoff, Booth, US 2890116 (1959 to U.S.A.). Structure: Bickoff et al., J. Am. Chem. Soc. 80, 3969 (1958). Synthesis: Emerson, Bickoff, ibid. 4381; US 2884427 (1959 to U.S.A.); Jurd, Tetrahedron Lett. 1963, 1151; Kappe, Brandner, Z. Naturforsch. 29B, 292 (1974). Biosynthesis: Grisebach, Barz, Chem. Ind. (London) 1963, 690.
Properties
Crystals, mp 385°. Sublimes at 325°; sublimes in high vacuum at about 175°. uv max (methanol): 208, 243, 343 nm. Exhibits bright blue fluorescence in neutral or acid soln, greenish-yellow fluorescence in strong alkali. Absorption and fluorescence spectra: O. S. Wolfbeis, K. Schaffner, Photochem. Photobiol. 32, 143 (1980). Practically insol in water at acid and neutral pH, in petr ether. Sparingly sol in water at alkaline pH (pH 11-12); slightly sol in methanol, chloroform, ether; very slightly sol in carbon tetrachloride, benzene.Derivative
Diacetate.C19H12O7; mol wt 352.29.
C 64.78%, H 3.43%, O 31.79%.
Properties
Crystals from acetic acid, mp 237°.Derivative
Dimethyl ether.C17H12O5; mol wt 296.27.
C 68.92%, H 4.08%, O 27.00%.