2566. Crabtree's Catalyst
Nomenclature
Description and references
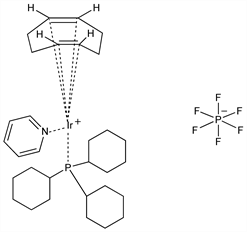
Stereoselective organoiridium based catalyst used primarily for hydrogenation of the general formula [Ir(cod)L(py)]metal complex where cod is 1,5-cyclooctadiene, L is tertiary phosphine, py is pyridine. Prepn: R. H. Crabtree, G. E. Morris, J. Organomet. Chem. 135, 395 (1977); R. H. Crabtree et al., ibid. 141, 205 (1977); R. H. Crabtree, S. M. Morehouse, Inorg. Synth. 24, 173 (1986). Hydrogenation study: J. W. Suggs et al., Tetrahedron Lett. 22, 303 (1981); directing effects: J. M. Brown, S. A. Hall, Tetrahedron 41, 4639 (1985); R. H. Crabtree, M. W. Davis, J. Org. Chem. 51, 2655 (1986). Structure: M. S. Abbassioun et al., Acta Crystallogr. C45, 331 (1989). Synthetic applications: M. Weck et al., J. Org. Chem. 64, 5463 (1999); J. M. Bueno et al., Tetrahedron Lett. 41, 4379 (2000).