2589. Crocetin
Nomenclature
CAS number: 27876-94-4
8,8′-Diapo-ψ,ψ-carotenedioic acid; trans-crocetin. C20H24O4; mol wt 328.40.
C 73.15%, H 7.37%, O 19.49%.
Description and references
Carotenoid-dicarboxylic acid isolated from Crocus sativus L.; C. albiflorus var. neapolitanus Hort.; C. luteus Lam., Iridaceae. Extraction procedure and structure: Jucker, Karrer, Carotinoide (Basel, 1948) p 282.
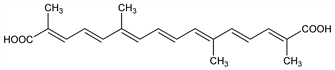
Properties
Brick-red rhombs from acetic anhydride, mp 285°. Absorption max (pyridine): 464, 436, 411 nm. Sol in pyridine, in very dil NaOH solns. Very sparingly sol in water, and in the usual organic solvents except pyridine and similar organic bases. Forms a solid dipyridyl salt.Derivative
Dimethyl ester.C22H28O4; mol wt 356.46.
C 74.13%, H 7.92%, O 17.95%.
Properties
Brick-red elongated leaflets, mp 222.5°. Total synthesis: Buchta, Andree, Naturwissenschaften 46, 74 (1959).Derivative
Di-gentiobiose ester.Nomenclature
CAS number: 42553-65-1
Crocin. C44H64O24; mol wt 976.96.
C 54.09%, H 6.60%, O 39.30%.
Description and references
One of the yellow-red pigments of saffron. Isoln and structure: Karrer, Salomon, Helv. Chim. Acta 10, 397 (1927); 11, 513, 711 (1928); Karrer et al., ibid. 12, 985 (1929); 13, 392 (1930); Kuhn, Winterstein, Ber. 67, 344 (1934); Reichstein, Angew. Chem. 74, 887 (1962).