2602. Crown Ethers
Nomenclature
Crown compounds.Description and references
Macrocyclic polyethers with the repeating unit (‐CH2‐CH2‐O‐)n, where n is greater than 2. Crown compounds with other heteroatoms (N, S, P) are known. Prepd by C. J. Pedersen, J. Am. Chem. Soc. 89, 2495, 7017 (1967). Described as “crown” ethers due to appearance of space-filling models and ability to “crown” cations. Proposed nomenclature lists non-ring substituents, number of atoms in ring, the class (crown), and the number of heteroatoms in the ring; e.g. dibenzo-18-crown-6. Bicyclic crowns are called cryptates. Crowns complex with cations and solubilize inorganic reagents in organic solvents. Selectivity results from the definite size of the crown cavity which admits only cations of corresponding ionic radii. The stability of complexes depends on the size of the cation and the size of the polyether ring. Chiral crowns are used in optical resolution of enantiomers. Reviews: D. J. Cram, J. M. Cram, Science 183, 803-809 (1974); eidem, Acc. Chem. Res. 11, 8-14 (1978); J. J. Christensen et al., Chem. Rev. 74, 351-384 (1974); G. W. Gokel, H. D. Durst, Synthesis 1976, 168-184; A. C. Knipe, J. Chem. Educ. 53, 618-622 (1976); V. Prelog, Pure Appl. Chem. 50, 893-904 (1978). Historical overview: C. J. Pedersen, Science 241, 536-540 (1988).
Derivative
18-Crown-6.Nomenclature
1,4,7,10,13,16-Hexaoxacyclooctadecane.Description and references
Prepn: G. W. Gokel et al., Org. Synth. 57, 30 (1977).
Properties
Crystals from acetonitrile, mp 38-39.5°.Derivative
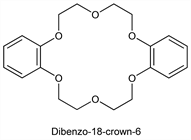
Dibenzo-18-crown-6.Nomenclature
2,3,11,12-Dibenzo-1,4,7,10,13,16-hexaoxacyclooctadeca-2,11-diene.Description and references
The first crown ether discovered. Prepn: C. J. Pedersen, US 3687978 (1972 to Du Pont).