2676. Curvularin
Nomenclature
CAS number: 10140-70-2
4,5,6,7,8,9-Hexahydro-11,13-dihydroxy-4-methyl-2H-3-benzoxacyclododecin-2,10(1H)-dione. C16H20O5; mol wt 292.33.
C 65.74%, H 6.90%, O 27.37%.
Description and references
Mold metabolite from a species of Convalaria: Musgrave, J. Chem. Soc. 1956, 4301; from Penicillium steckii: Fennell et al., Chem. Ind. (London) 1959, 1382. Structure: Birch et al., J. Chem. Soc. 1959, 3146. Synthesis: H. Gerlach, Helv. Chim. Acta 60, 3039 (1977); of dl-O,O-dimethyl ether: Baker et al., J. Chem. Soc. C 1967, 1913; T. Takahashi et al., Tetrahedron Lett. 21, 3885 (1980); H. H. Wasserman, R. J. Gambale, ibid. 22, 4849 (1981).
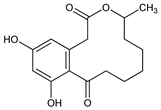
Properties
Plates from hot benzene + methanol, mp 206-206.5°. [α]D18 -36.3° (c = 3.8 in ethanol). uv max (ethanol): 223, 272, 304.5 nm (ε 11300; 6350; 5100). Sol in ethanol, methanol, dioxane, acetone, pyridine, concd H2SO4; moderately sol in acetic acid, ether; sparingly sol in benzene, petr ether, chloroform, water. Gives yellow solns with aq ammonia, sodium carbonate, and sodium hydroxide. Alkaline solns darken rapidly in air, eventually becoming purple.Derivative
Dibenzoate.C30H28O7; mol wt 500.54.
C 71.99%, H 5.64%, O 22.37%.
Properties
Needles from benzene + petr ether, mp 133-134°. [α]D18 -10.8° (c = 1.9 in chloroform). uv max (ethanol): 236 nm (ε 36700).Derivative
O,O-Dimethyl ether.C18H24O5; mol wt 320.38.
C 67.48%, H 7.55%, O 24.97%.