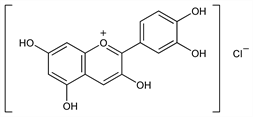
2687. Cyanidin Chloride
Nomenclature
CAS number: 528-58-5
2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-1-benzopyrylium chloride; 3,3′,4′,5,7-pentahydroxyflavylium chloride; 3,3′,4′,5,7-pentahydroxy-2-phenylbenzopyrylium chloride. C15H11ClO6; mol wt 322.70.
C 55.83%, H 3.44%, Cl 10.99%, O 29.75%.
Description and references
Prepd by acid hydrolysis of cyanin chloride: Willst"atter, Everest, Ann. 401, 189 (1913). Isoln from bananas: Simmonds, Nature 173, 402 (1954). Prepn by reduction of quercetin: Bauer et al., Chem. Ind. (London) 1954, 433; King, White, J. Chem. Soc. 1957, 3901. Structure: Willst"atter, Mallison, Ann. 408, 147 (1915). Synthesis: Willst"atter et al., Ber. 57, 1938 (1924); Robertson, Robinson, J. Chem. Soc. 1928, 1528. Biosynthesis: Fritsch et al., Z. Naturforsch. 26b, 581 (1971). See also Bioflavonoids.
Properties
Metallic brownish-red needles (monohydrate) from dil alcoholic HCl. The anhydr compd does not melt below 300°. Absorption max (methanolic HCl): 535 nm. Freely sol in alcohol and in amyl alcohol giving a violet soln. Sol in sodium carbonate soln with blue color. Sparingly sol in dil HCl or H2SO4.Derivative
3-Glucoside.Nomenclature
Chrysanthemin; asterin; kuromanin.C21H21ClO11; mol wt 484.84.
C 52.02%, H 4.37%, Cl 7.31%, O 36.30%.
Properties
From winter aster (Chrysanthemum indicum L., Compositae): Willst"atter, Bolton, Ann. 412, 136 (1917); from wild strawberries (Fragaria vesca L., Rosaceae): Sondheimer, Karash, Nature 178, 648 (1956); from sweet cherries (Prunus avium L., Rosaceae): Li, Wagenknecht ibid. 182, 657 (1958). Identity with asterin: Robinson, Willst"atter, Ber. 61, 2503 (1928). Structure and synthesis: Murakami et al., J. Chem. Soc. 1931, 2665. Reddish-brown plates or prisms with a metallic shine from dil alcoholic HCl, dec 205° without melting. Absorption max (methanolic HCl): 525 nm. Sol in alc with strong fluorescence and in sodium carbonate with violet-blue color.Derivative
3-Rhamnoglucoside.Nomenclature
Keracyanin; antirrhinin; sambucin; Meralop (ISF); Meralops (Dulcis).C27H31ClO15; mol wt 630.98.
C 51.39%, H 4.95%, Cl 5.62%, O 38.03%.
Properties
From skin of sweet cherries (Prunus avium L,. Rosaceae): Willst"atter, Zollinger, Ann. 412, 164 (1917); from sour cherries (Prunus cerasus L., Rosaceae): Li, Wagenknecht, J. Am. Chem. Soc. 78, 979 (1956). Structure: Robertson, Robinson, J. Chem. Soc. 1927, 2196. Prepn by reduction of quercetin-3-rhamnoglucoside: Bauer et al., Chem. Ind. (London) 1954, 433. Reduces time to adjust to darkness: F. Trimarchi et al., Minerva Oftalmol. 18, 143 (1977). Mechanism of action: F. Trimarchi et al., Ann. Ottalmol. Clin. Ocul. 105, 111 (1979). Red needles from dil HCl or dark prisms from dil methanolic HCl. Absorption max (ethanolic HCl): 532, 333, 282 nm. Sol in hot water, alcohol.Derivative
3-Galactoside.Nomenclature
Idaein; idein.C21H21ClO11; mol wt 484.84.
C 52.02%, H 4.37%, Cl 7.31%, O 36.30%.
Properties
From cranberries (Vaccinium vitis idaea L., Ericaceae): Willst"atter, Mallison, Ann. 408, 15 (1915); from Winesap apple (Pyrus malus Linn., Rosaceae): Duncan, Dustman, J. Am. Chem. Soc. 58, 1511 (1936). Structure and synthesis: Grove, Robinson, J. Chem. Soc. 1931, 2722. Red needles with bronze metallic luster from dil ethanolic HCl, dec 210-212°. Sol in water, ethanol, methanol, dil HCl. Practically insol in 7% HCl.Derivative
3,5-Diglucoside.Nomenclature
Cyanin; shisonin A.C27H31ClO16; mol wt 646.98.
C 50.12%, H 4.83%, Cl 5.48%, O 39.57%.
Properties
From cornflower (Centaurea cyanus L., Compositae): Willst"atter, Everest, loc. cit. Structure: Léon, Robinson, J. Chem. Soc. 1932, 221. Synthesis: Robinson, Todd, ibid. 2488. Plates with a metallic luster from dil alcoholic HCl, mp 203-204°. [α]D -258° (in 0.05% HCl). Absorption max (methanolic HCl): 522 nm.Derivative
3-Sophoroside.Nomenclature
Mecocyanin.C27H31ClO16; mol wt 646.98.
C 50.12%, H 4.83%, Cl 5.48%, O 39.57%.
Properties
From the flowers of Papaver rhoeas L., Papaveraceae: Willst"atter, Weil, Ann. 412, 231 (1917); from sour cherries: Li, Wagenknecht, J. Am. Chem. Soc., loc. cit. Structure: Harborne, Experientia 19, 7 (1963); Phytochemistry 2, 85 (1963). (Alternate structure: Grove et al., J. Chem. Soc. 1934, 1608). Dark-red crystals from dil HCl + HOAc or dark-red needles from 2% alcoholic HCl. Absorption max (methanolic HCl): 523 nm. Sol in water. Pptd by glacial acetic acid or acetone.Therapeutic Category
3-Rhamnoglucoside in treatment of night blindness.