2726. Cyclohexanone
Nomenclature
CAS number: 108-94-1
Ketohexamethylene; pimelic ketone; Hytrol O; Anone; Nadone. C6H10O; mol wt 98.14.
C 73.43%, H 10.27%, O 16.30%.
Description and references
Obtained from cyclohexanol by catalytic dehydrogenation or by oxidation (which yields cyclohexanone and adipic acid) or from cyclohexane by oxidation (yielding cyclohexanone and cyclohexanol): GB 310055 (1928 to Schering-Kahlbaum); US 2223493; US 2223494; US 2285914 (1940, 1940, 1942 to Du Pont). Review of manuf processes: Faith, Keyes & Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp 304-309.
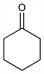
Properties
Oily liquid with characteristic odor reminiscent of peppermint and acetone. Flammable. d420 0.9478; d425 0.9421. mp -32.1°. bp760 155.6°; bp400 132.5°; bp200 110.3°; bp100 90.4°; bp60 77.5°; bp40 67.8°; bp20 52.5°; bp10 38.7°; bp5 26.4°; bp1.0 1.4°. nD20 1.4507. Flash pt (closed cup): 111°F (44°C). Soly in water: 150 g/l at 10°; 50 g/l at 30°. Soly of water in cyclohexanone: 87 g/l at 20°. Sol in alcohol, ether and in other common organic solvents. LD50 orally in rats: 1.62 ml/kg. See: H. F. Smyth et al., Am. Ind. Hyg. Assoc. J. 30, 470 (1969).Derivative
2,4-Dinitrophenylhydrazone.Nomenclature
CAS number: 1589-62-4
2-(2,4-Dinitrophenyl)hydrazone cyclohexanone. C12H14N4O4; mol wt 278.26.
C 51.80%, H 5.07%, N 20.13%, O 23.00%.