2737. Cyclonium Iodide
Nomenclature
CAS number: 6577-41-9
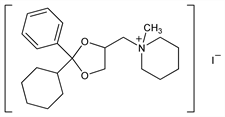
1-[(2-Cyclohexyl-2-phenyl-1,3-dioxolan-4-yl)methyl]-1-methylpiperidinium
iodide; N-methyl-N-[(2-cyclohexyl-2-phenyl-1,3-dioxolan-4-yl)-methyl]piperidinium
iodide; 1-(2-phenyl-2-cyclohexyl-1,3-dioxolan-4-yl)methyl-1-methylpiperidinium
iodide; ciclonium iodide; oxapium iodide; SH-100; Esperan (Toyama). C22H34INO2; mol wt 471.42.
C 56.05%, H 7.27%, I 26.92%, N 2.97%, O 6.79%.
Description and references
Prepn: M. Kimura et al., JP 66 6585 (1966 to Toyama), C.A. 65, 7191 (1966); and separation of isomers: I. Saikawa, JP 70 16475 (1970 to Toyama), C.A. 73, 45355t. Stability studies: Shiuchi, Miyazaki, Bunseki Kagaku 18, 455 (1969), C.A. 71, 74097z (1969). Incompatibility studies: Kunita et al., Yakuzaigaku 28, 84 (1968), C.A. 69, 99335x (1968). Anticholinergic activity and acute toxicity: J. Faff et al., Pol. J. Pharmacol. Pharm. 30, 493 (1978). Review: Jpn. Med. Gaz. 8(9), 11 (1971).
Derivative
α-Form.Properties
White crystals from monochlorobenzene or isopropyl alcohol, mp 195-197°. Insol in trichloroethylene.Derivative
β-Form.Properties
White crystals from monochlorobenzene, mp 150-152°. Sol in hot trichloroethylene. Both sol in methanol, ethanol, chloroform and tetrachlorethane. Hardly sol in benzene, toluene, xylene and water. LD50 i.p. in mice: 106 mg/kg (Faff).Therapeutic Category
Antispasmodic.
Keywords
Antispasmodic