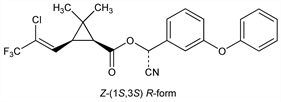
2758. λ-Cyhalothrin
Nomenclature
Description and references
Synthetic pyrethroid; 1:1 mixture of the Z-(1R,3R) S-ester isomers and Z-(1S,3S) R-ester isomers of cyhalothrin, q.v. Prepn: M. J. Robson, J. Crosby, EP 106469; J. Crosby, US 4510098 (1984, 1985 both to ICI); M. J. Robson et al., Brighton Crop Prot. Conf. - Pests Dis. 1984, 853. Description of chemical properties and biological activities: A. R. Jutsum et al., ibid. 421. Field trials for mosquito control: A. A. Weathersbee et al., J. Am. Mosq. Control Assoc. 7, 238 (1991); for sandfly control: D. A. Elnaiem et al., Med. Vet. Entomol. 13, 310 (1999). Effect on beneficial insects: J. S. White et al., Brighton Crop Prot. Conf. - Pests Dis. 1990, 969. Dissipation on soil: B. D. Hill, D. J. Inaba, J. Agric. Food Chem. 39, 2282 (1991). Determination in natural waters: S. T. Hadfield et al., Pestic. Sci. 34, 207 (1992). Review of early field trials on horticultural crops: D. Wilson, J. Trevenna, Brighton Crop Prot. Conf. - Pests Dis. 1986, 323.