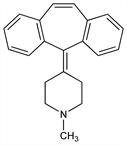
2773. Cyproheptadine
Nomenclature
Description and references
Serotonin and histamine antagonist. Prepn: E. L. Engelhardt, US 3014911 (1961 to Merck & Co.); E. L. Engelhardt et al., J. Med. Chem. 8, 829 (1965). Improved prepn: M. M. Cid et al., Tetrahedron 44, 6197 (1988). Metabolism: C. C. Porter et al., Drug Metab. Dispos. 3, 189 (1975). HPLC determn in serum and plasma: E. A. Novak et al., J. Chromatogr. 339, 457 (1985). Toxicity study: J. J. Loux et al., Arzneim.-Forsch. 28, 1644 (1978). Comprehensive description: H. Y. Aboul-Enein, A. A. Al-Badr, Anal. Profiles Drug Subs. 9, 155-179 (1980). Clinical trial as an appetite stimulant in cystic fibrosis: D. N. Homnick et al., Pediatr. Pulmonol. 40, 251 (2005).
Properties
Crystals from dil ethanol, mp 112.3-113.3°.Derivative
Hydrochloride sesquihydrate.Nomenclature
Properties
Crystals from abs ethanol + ether, dec 252.6-253.6°. uv max (0.1N H2SO4): 224, 285 nm (E1%1cm 1656, 355). 1.0 g is sol in 1.5 ml methanol, 16 ml chloroform, 35 ml alc, 275 ml H2O. Practically insol in ether. LD50 orally in mice: 74.2 mg/kg (Loux).Derivative
Hydrochloride monohydrate.Nomenclature
Properties
Crystals, mp 214-216°. Soly in water: ≈5 mg/ml.Therapeutic Category
Antihistaminic; antipruritic; appetite stimulant.
Therapeutic Category (Veterinary)
Antihistaminic; antipruritic; appetite stimulant.Keywords
Antihistaminic; Tricyclics; Other Tricyclics; Antipruritic