2796. 2,4-D
Nomenclature
CAS number: 94-75-7
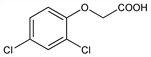
(2,4-Dichlorophenoxy)acetic acid; Trinoxol (Crystal Chem.). C8H6Cl2O3; mol wt 221.04.
C 43.47%, H 2.74%, Cl 32.08%, O 21.71%.
Description and references
Prepd from 2,4-dichlorophenol and monochloroacetic acid in aq NaOH: Pokorny, J. Am. Chem. Soc. 63, 1768 (1941); Foster, GB 573476 (1945); by chlorination of molten phenoxyacetic acid: Manske, US 2471575 (1949 to U.S. Rubber); from 2,4-dichlorophenol, sodium, and ethyl chloroacetate followed by hydrolysis of the ester: Haskelberg, J. Org. Chem. 12, 426 (1947). Toxicology: V. K. Rowe, T. A. Hymas, Am. J. Vet. Res. 15, 622 (1954); J. M. Way, Residue Rev. 26, 37 (1969).
Properties
Crystals from benzene, mp 138°. bp0.4 160°. Almost insol in water. Sol in organic solvents. LD50 in mice, rats (mg/kg): 368, 375 orally (Rowe, Hymas).Derivative
Sodium salt.C8H5Cl2NaO3; mol wt 243.02.
C 39.54%, H 2.07%, Cl 29.18%, Na 9.46%, O 19.75%.
Properties
Needles from alc, dec 215°. Soly in water: 3.5%.Derivative
Isopropyl ester.Nomenclature
Esteron 44 (Dow).C11H12Cl2O3; mol wt 263.12.
C 50.21%, H 4.60%, Cl 26.95%, O 18.24%.
Properties
Liquid, d2525 1.255-1.270. Solidifies at +5°. Sol in oil.Derivative
Butyl ester.Nomenclature
Lironox; Weedone (Nufarm).C12H14Cl2O3; mol wt 277.14.
C 52.01%, H 5.09%, Cl 25.58%, O 17.32%.
Description and references
Prepn: Nagel, DE 1144288 (1963 to Bayer), C.A. 59, 6315e (1963).