2800. Dactinomycin
Nomenclature
Description and references
Antibiotic substance belonging to the actinomycin complex, produced by several Streptomyces spp. Historical background of actinomycins and chemistry, toxicology, pharmacology of actinomycin D: Ann. N.Y. Acad. Sci. 89, 285-485 (1960). Isoln from broth cultures of S. parvulus: Manaker et al., Antibiot. Ann. 1954/55, 853. Structure: Bullock, Johnson, J. Chem. Soc. 1957, 3280. Synthesis: Brockmann, Manegold, Naturwissenschaften 51, 383 (1964); Brockmann, Lackner, ibid. 384, 435 (1964); Brockmann et al., DE 1172680 (1964 to Bayer); eidem, Ber. 101, 1312 (1968); Meienhofer, J. Am. Chem. Soc. 92, 3771 (1970); T. Tanaka et al., Bull. Chem. Soc. Jpn. 53, 1352 (1980); K. Nakajima et al., Pept. Chem. 19, 143-148 (1981). Conformation from NMR: Conti, DeSantis, Nature 227, 1239 (1970); Lackner, Ber. 104, 3653 (1971). Toxicity data: F. S. Philips et al., Ann. N.Y. Acad. Sci. 89, 348 (1960). Mechanism of action study: P. Delay-Goyet, J. M. Lundberg, Biochem. Biophys. Res. Commun. 180, 1342 (1991). Clinical evaluation in Wilms' tumor: B. de Camargo, E. L. Franco, Cancer 73, 3081 (1993). Review and evaluation of studies of carcinogenic action in laboratory animals: IARC Monographs 12, 29-41 (1976).
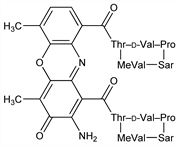
Derivative
Trihydrate.Properties
Bright red, rhomboid prisms from abs alc, dec 241.5-243°. [α]D28 -315° (c = 0.25 in methanol). Abs max (methanol): 244, 441 nm (A1%1cm 281, 206). Dil solns are very sensitive to light. Sol in alc, propylene glycol, water + glycol mixtures. LD50 orally in mice, rats: 13.0, 7.2 mg/kg (Philips).Therapeutic Category
Antineoplastic.
Therapeutic Category (Veterinary)
Antineoplastic.Keywords
Antineoplastic; Antibiotics and Analogs; Actinomycins