2804. Dalfopristin
Nomenclature
Description and references
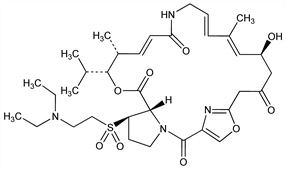
Semisynthetic polyunsaturated macrolactone type II streptogramin, q.v. Prepn: J.-C. Barriere et al., EP 191662; eidem, US 4668669 (1986, 1987 both to Rhone-Poulenc). In vitro activity: H. C. Neu et al., J. Antimicrob. Chemother. 30, Suppl. A, 83 (1992). HPLC determn in plasma: A. Le Liboux et al., J. Chromatogr. B 708, 161 (1998)
Properties
White solid, mp ≈150°.Derivative
Mixture with quinupristin.Nomenclature
Description and references
Semisynthetic streptogramin comprised of two synergistic components in a defined 70:30 percent w/w mixture of dalfopristin and quinupristin, q.v., mesylate salts. HPLC determn for quality control: B. Vasselle et al., J. Pharm. Biomed. Anal. 19, 641 (1999). In vitro activity in comparison with pristinamycin, q.v.: A. Lozniewski et al., Pathol. Biol. 48, 463 (2000). Clinical trial in vancomycin resistant Enterococcus faecium (VREF) infection: R. C. Moellering et al., J. Antimicrob. Chemother. 44, 251 (1999); in skin infections: R. L. Nichols et al., ibid. 263. Review: B. Pavan, Curr. Opin. Investig. Drugs 1, 173-180 (2000).
Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics)