2816. Dantrolene
Nomenclature
Description and references
Direct acting skeletal muscle relaxant; probably inhibits the release of calcium from the sarcoplasmic reticulum of skeletal muscle. Prepn: NL 6612588; Davis, Snyder, US 3415821 (1967, 1968 both to Norwich Pharmacal); Snyder et al., J. Med. Chem. 10, 807 (1967); Frimm et al., Chem. Zvesti 23, 916 (1969). HPLC determn in plasma: M. Lalande et al., J. Chromatogr. 430, 187 (1988). Toxicity data: K. O. Ellis et al., J. Pharm. Sci. 69, 327 (1980). Review of pharmacology: G. G. Harrison, Br. J. Anaesth. 60, 279 (1988); and therapeutic use: A. Ward et al., Drugs 32, 130-168 (1986). Review of efficacy in malignant hyperthermia: R. Ben Abraham et al., Q. J. Med. 90, 13-18 (1997); of pharmacology and clinical indications: T. Krause et al., Anaesthesia 59, 364-373 (2004).
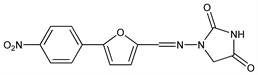
Properties
Crystals from aqueous DMF, mp 279-280°.Derivative
Sodium salt hemiheptahydrate.Nomenclature
Properties
Orange powder. Slightly soluble in water; more sol in alkaline soln. pKa 7.5. LD50 (calculated as base) orally in mice: 1110 mg/kg (Ellis).Therapeutic Category
Muscle relaxant (skeletal); in treatment of malignant hyperthermia.
Therapeutic Category (Veterinary)
Muscle relaxant (skeletal).Keywords
Muscle Relaxant (Skeletal)