2819. Daphnoline
Nomenclature
CAS number: 479-36-7
6,6′-Dimethoxy-2-methyloxyacanthan-7,12′-diol; trilobamine. C35H36N2O6; mol wt 580.67.
C 72.39%, H 6.25%, N 4.82%, O 16.53%.
Description and references
From bark of Daphnandra micrantha Benth., Monimiaceae. Isoln: Pyman, J. Chem. Soc. 105, 1679 (1914). Identity with trilobamine: Bick et al., ibid. 1949, 2767. Structure: Bick et al., ibid. 1960, 4928.
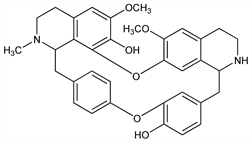
Properties
Crystals from chloroform, mp 194-196°. [α]D +459° (c = 0.3 in chloroform). uv max (methanol): 285 nm (log ε 3.9). Practically insol in water, ethyl acetate, acetone, ether, petr ether. Sparingly sol in methanol, ethanol, xylene, hot chloroform sol in dil acids, cold aq 5% sodium hydroxide.Derivative
Daphnandrine.Nomenclature
CAS number: 1183-76-2
6,6′,12′-Trimethoxy-2-methyloxyacanthan-7-ol. C36H38N2O6; mol wt 594.70.
C 72.71%, H 6.44%, N 4.71%, O 16.14%.