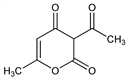
2865. Dehydroacetic Acid
Nomenclature
CAS number: 520-45-6
3-Acetyl-6-methyl-2H-pyran-2,4(3H)-dione; 2-acetyl-5-hydroxy-3-oxo-4-hexenoic acid δ-lactone; methylacetopyronone; DHA. C8H8O4; mol wt 168.15.
C 57.14%, H 4.80%, O 38.06%.
Description and references
Polymerization product of ketene: Steele et al., J. Org. Chem. 14, 460 (1949). From ethyl acetoacetate: Arndt, Org. Synth. coll. vol. III, 231 (1955). Toxicity study: H. C. Spencer et al., J. Pharmacol. Exp. Ther. 99, 57 (1950). Series of articles on toxicity studies, pharmacology, mechanism of action, absorption, distribution, renal action: ibid. 57-111.
Properties
White to cream crystalline powder, mp 109-111° (sublimes). bp 269.9°. Soly (w/w at 25°): in acetone 22%; benzene 18%; methanol 5%; carbon tetrachloride 3%; U.S.P. ethanol 3%; ether 5%; glycerol <0.1%, n-heptane 0.7%; olive oil 1.6%; propylene glycol 1.7%; water <0.1%. LD50 orally in rats: 1000 mg/kg (Spencer).Derivative
Sodium salt hydrate.Nomenclature
DHA-S (Dow).C8H7NaO4.H2O; mol wt 208.14.
C 46.16%, H 4.36%, Na 11.05%, O 38.43%.