2871. Dehydroepiandrosterone
Nomenclature
Description and references
Major secretory steroidal product of the adrenal gland; secretion progressively declines with aging. May have estrogen- or androgen-like effects depending on the hormonal milieu. Intracellularly converted to androstenedione, q.v. Isoln from male urine: Butenandt, Tscherning, Z. Physiol. Chem. 229, 167 (1934); Butenandt, Dannenbaum, ibid. 192. Prepn from cholesterol: Butenandt et al., ibid. 237, 57 (1935); Ruzicka, Wettstein, Helv. Chim. Acta 18, 986 (1935); Wallis, Fernholz, J. Am. Chem. Soc. 57, 1379, 1504 (1935); Schoeller et al., Naturwissenschaften 23, 337 (1935); from sitosterol: Oppenauer, Nature 135, 1039 (1935). High yield prepn: H. Hosoda et al., J. Org. Chem. 38, 4209 (1973). Metabolism study: P. Knapstein et al. Acta Endocrinol. 58, 261 (1968). Toxicity study of the sulfate: M. Yahara et al., J. Toxicol. Sci. 2, 161 (1977). Review of physiological importance: P. Ebeling, V. A. Koivisto, Lancet 343, 1479-1481 (1994). Symposium on role in aging: Ann. N.Y. Acad. Sci. 774, 1-350 (1995). Review of therapeutic potential in aging: B. Allolio, W. Arlt, Trends Endocrinol. Metab. 13, 288-294 (2002); of clinical experience in lupus: P. Kocis, Am. J. Health Syst. Pharm. 63, 2201-2210 (2006).
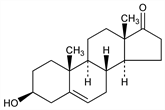
Properties
Dimorphous, needles, mp 140-141°; leaflets, mp 152-153°. [α]18D +10.9° (c = 0.4 in alc). Pptd by digitonin. Sol in benzene, alcohol, ether; sparingly sol in chloroform, petr ether.Derivative
Sulfate.Nomenclature
Description and references
Hydrophilic storage form that circulates in the blood.
Derivative
Sodium sulfate.Nomenclature
Properties
White cryst powder, mp 154° (dec). Sol in methanol, slightly sol in water, abs ethanol. Practically insol in acetone, chloroform, benzene.Therapeutic Category
In treatment of menopausal syndrome and systemic lupus erythematosus (SLE).
Keywords
Androgen