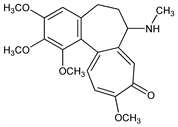
2887. Demecolcine
Nomenclature
CAS number: 477-30-5
6,7-Dihydro-1,2,3,10-tetramethoxy-7-(methylamino)benzo[a]heptalen-9(5H)-one; N-deacetyl-N-methylcolchicine; N-methyl-N-desacetylcolchicine; colchamine; Santavy's substance F; colcemid. C21H25NO5; mol wt 371.43.
C 67.91%, H 6.78%, N 3.77%, O 21.54%.
Description and references
Antitumor alkaloid; depolymerizes microtubules and inhibits spindle formation during metaphase. Isoln from Colchicum autumnale L., Liliaceae: Santavy, Pharm. Acta Helv. 25, 248 (1950); Santavy, Reichstein, Helv. Chim. Acta 33, 1606 (1950); Schlittler, Uffer, DE 936268 (1955 to Ciba), C.A. 53, 1396 (1959). Structure: Santavy et al., Helv. Chim. Acta 36, 1319 (1953). Synthesis: Uffer et al., ibid. 37, 18 (1954); H. G. Capraro, A. Brossi, ibid. 62, 965 (1979). Effect on the mitotic cycle: C. L. Rieder, R. E. Palazzo, J. Cell Sci. 102, 387 (1992). Use in oocyte enucleation: E. Ibá~nez et al., Biol. Reprod. 68, 1249 (2003).