2929. Desosamine
Nomenclature
CAS number: 5779-39-5
3,4,6-Trideoxy-3-(dimethylamino)-d-xylo-hexose; 4-dimethylaminotetrahydro-6-methylpyran-2,3-diol; picrocine. C8H17NO3; mol wt 175.23.
C 54.83%, H 9.78%, N 7.99%, O 27.39%.
Description and references
Sugar component of several macrolide antibiotics, such as the erythromycins. Isoln: Clark, Antibiot. Chemother. 3, 663 (1953). Identity with picrocine: Brockmann et al., Ber. 87, 856 (1954). Stereochemistry: Woo et al., Tetrahedron Lett. 1962, 735. Configuration: Bolton et al., Chem. Ind. (London) 1962, 1945; Foster et al., J. Chem. Soc. 1965, 2318. Synthesis of dl-form: Korte et al., Tetrahedron 18, 657 (1962); Newman, Chem. Ind. (London) 1963, 372; J. Org. Chem. 29, 1461 (1964). Stereospecific synthesis of the natural d-form: Richardson, J. Chem. Soc. 1964, 5364; of the l-form: Baer, Chiu, Can. J. Chem. 52, 122 (1974).
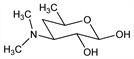
Properties
Crystals, mp 86-87°.Derivative
Hydrochloride.C8H17NO3.HCl; mol wt 211.69.
C 45.39%, H 8.57%, N 6.62%, O 22.67%, Cl 16.75%.