Nomenclature
CAS number: 123-42-2
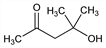
4-Hydroxy-4-methyl-2-pentanone; Pyranton.
C
6H
12O
2; mol wt 116.16.
C 62.04%, H 10.41%, O 27.55%.
Description and references
Prepd by the action of barium hydroxide or
of calcium hydroxide on acetone: Conant, Tuttle, Org. Synth. vol. 1, p 45 (1921); coll.
vol. I, 193; cf. Jacquemain, Compt. Rend. 196, 1622 (1933). Industrial
process using KOH and acetone: Schmitt, Disteldorf, DE 1052970 (1959 to Hibernia). Toxicity study: Smyth, Carpenter, J. Ind. Hyg. Toxicol. 30, 63 (1948).
Properties
Liquid with faint, pleasant odor. d425 0.9306 (0.940 for tech grades). bp760 167.9°. bp100 108.2°. bp20 72.0°. bp10 58.8°. bp1.0 22.0°. mp -44°; nD20 1.4232: Lantz, J. Am. Chem. Soc. 62, 3260 (1940). Flash pt
reagent grade: 66°C (151°F). Flash pt commercial grades: 8°C (48°F) (closed cup); 13°C (55°F) (open cup). Vapor density 4.00 (air = 1.00). Flammable. Miscible with water, alcohol, ether, and other solvents. Dec by
prolonged exposure to alkalies and by distillation at atmospheric
pressure. LD50 orally in rats: 4.0 g/kg (Smyth).Caution
Potential symptoms of overexposure
are irritation of eyes, nose, throat and skin; corneal damage. See NIOSH Pocket Guide to Chemical
Hazards (DHHS/NIOSH 97-140, 1997) p 90.Use
Solvent for cellulose acetate, nitrocellulose, celluloid,
fats, oils, waxes, resins. As a preservative in pharmaceutical prepns.
In some antifreeze solns and in hydraulic fluids.
