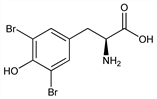
3030. 3,5-Dibromo-l-tyrosine
Nomenclature
CAS number: 300-38-9
β-(3,5-dibromo-4-hydroxyphenyl)alanine; Biotiren (Benvegna); Bromotiren. C9H9Br2NO3; mol wt 338.98.
C 31.89%, H 2.68%, Br 47.14%, N 4.13%, O 14.16%.
Description and references
Obtained upon saponification of gorgonin, a substance isolated from coral (Primnoa lepadifera) stems: M"orner, Z. Physiol. Chem. 88, 139, 152 (1913). Prepd by the action of bromine vapor on tyrosine: Gorup-Besanez, Ann. 125, 281 (1863). By treating tyrosine in aq HBr with Br: Aloy, Rabaut, Bull. Soc. Chim. Fr. [4] 3, 392 (1908); Zeynek, Z. Physiol. Chem. 114, 275 (1921). Clinical evaluation in thyroid function: A. Isidori, Int. J. Clin. Pharmacol. Biopharm. 16, 180 (1978).
Derivative
l-Form dihydrate.Properties
Efflorescent needles, plates (orthorhombic) from water. When anhydr, dec 245°. [α]D20 +1.3° (c = 5 in 4% HCl). pK1 2.17; pK2 6.45; pK3 7.60. One gram dissolves in 250 ml water at 25°, in 30 ml at boiling temp. Slightly sol in alcohol; insol in ether. Freely sol in alkalies and in dil mineral acids. Forms HBr and HCl salts; stable in boiling water.Derivative
dl-Form.Properties
Efflorescent prisms, platelets from water probably contg 1 mol water of crystn. Dec at about 245°. Aq solns are acid to litmus. Behaves as a monobasic acid when titrated with NaOH and phenolphthalein. One gram dissolves in about 590 ml water at 20°.Therapeutic Category
Thyroid inhibitor.
Keywords
Antihyperthyroid