3084. Dicloxacillin
Nomenclature
CAS number: 3116-76-5
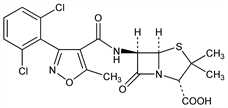
(2S,5R,6R)-6-[[[3-(2,6-Dichlorophenyl)-5-methyl-4-isoxazolyl]carbonyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; 6-[3-(2,6-dichlorophenyl)-5-methyl-4-isoxazolecarboxamido]penicillanic
acid; 3-(2,6-dichlorophenyl)-5-methyl-4-isoxazolylpenicillin; BRL-1702; Maclicine (Christiaens). C19H17Cl2N3O5S; mol wt 470.33.
C 48.52%, H 3.64%, Cl 15.08%, N 8.93%, O 17.01%, S 6.82%.
Description and references
Semi-synthetic antibiotic related to penicillin. Prepn: J. H. C. Naylor, GB 978299; eidem, US 3239507 (1965, 1969 to Beecham). Toxicity data: C. Gloxhuber et al., Arzneim.-Forsch. 15, 322 (1965). Series of articles on chemistry, pharmacology and toxicology: ibid. 322-348; Matsuzaki, Jpn. J. Antibiot. 21, 274, 284 (1968), C.A. 70, 95267z, 95265x (1969).
Derivative
Sodium salt monohydrate.Nomenclature
CAS number: 13412-64-1
Sodium dicloxacillin monohydrate; P-1011; Brispen; Dichlor-Stapenor (Bayer); Diclocil (BMS); Dycill (SKB); Dynapen (Apothecon); Noxaben (Unifa); Pathocil (Wyeth); Pen-Sint (Cophar); Syntarpen (Polfa); Veracillin (Ayerst). C19H16Cl2N3NaO5S.H2O; mol wt 510.32.
C 44.72%, H 3.56%, Cl 13.89%, N 8.23%, Na 4.50%, O 18.81%, S
6.28%.
Properties
Crystals, dec 222-225°. [α]D20 +127.2° (water). Sol in water, methanol; less sol in butanol; slightly sol in acetone and the usual organic solvents. LD50 in mice (g/kg): 0.9 i.v.; in rats (g/kg): 0.63 i.p.; >5 orally (Gloxhuber).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins