3090. Dicumarol
Nomenclature
CAS number: 66-76-2
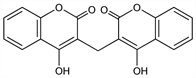
3,3′-Methylenebis[4-hydroxy-2H-1-benzopyran-2-one]; 3,3′-methylenebis[4-hydroxycoumarin]; bishydroxycoumarin (rescinded); Dicoumarol; Dicoumarin; Dicumol; Melitoxin. C19H12O6; mol wt 336.29.
C 67.86%, H 3.60%, O 28.55%.
Description and references
Originally isolated from spoiled sweet clover (improperly cured Melilotus hay): Link et al., J. Biol. Chem. 138, 21, 513, 529 (1941); 142, 941 (1942); now prepd synthetically by the action of formaldehyde on 4-hydroxycoumarin (obtained as the Na derivative by the action of sodium metal on methyl acetylsalicylate): Link, Fed. Proc. 4, 176 (1945), and patents (The Wisconsin Alumni Res. Found.). Toxicity data: Rose et al., Proc. Soc. Exp. Biol. Med. 50, 228 (1942).
Properties
Minute crystals. Slight pleasant odor. Has a slightly bitter taste. mp 287-293°. Sol in aq alkaline solns, in pyridine and similar organic bases. Slightly sol in benzene and chloroform. Practically insol in water, alcohol, ether. LD50 orally in rats: 541.6 mg/kg (Rose).Therapeutic Category
Anticoagulant.
Keywords
Anticoagulant