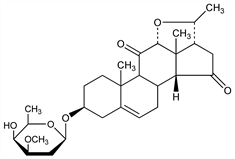
3157. Diginin
Nomenclature
CAS number: 467-53-8
(3β,12α,14β,17α,20S)-3-[(2,6-Dideoxy-3-O-methyl-d-lyxohexopyranosyl)oxy]-12,20-epoxypregn-5-ene-11,15-dione; 3β-(diginosyloxy)-12α,20α-epoxy-14β,17α-pregn-5-ene-11,15-dione. C28H40O7; mol wt 488.61.
C 68.83%, H 8.25%, O 22.92%.
Description and references
Isoln from leaves of Digitalis purpurea L., Scrophulariaceae: Karrer in E. Barell Festschrift (1936) p 238; Chem. Zentralbl. 1936, II, 2727. Isoln: Shoppee, Reichstein, Helv. Chim. Acta 23, 975 (1940); Shoppee, ibid. 27, 426 (1944). Structure: Shoppee et al., J. Chem. Soc. 1962, 3610; Tschesche, Brügmann, Tetrahedron 20, 1469 (1964). Mild hydrolysis yields diginigenin and diginose. Attempted partial synthesis of diginigenin: Tschesche, Schwinum, Ber. 100, 464 (1967); Tschesche, Müller-Albrecht, Ber. 103, 350 (1970).