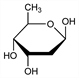
3164. Digitoxose
Nomenclature
Description and references
Obtained by mild acid hydrolysis of the glycosides digitoxin, gitoxin and digoxin: Cloetta, Arch. Exp. Pathol. Pharmakol. 88, 113 (1920); 112, 261 (1926); Windaus, Stein, Ber. 61, 2436 (1928); Kraft, Arch. Pharm. 250, 118 (1912); Mannich et al., ibid. 268, 453 (1930); Smith, J. Chem. Soc. 1930, 508; 1931, 23. Configuration: Micheel, Ber. 63, 347 (1930). Structure: S. F. Dyke, The Carbohydrates (Interscience, New York, 1960) p 104. Synthesis: Gut, Prins, Helv. Chim. Acta 30, 1223 (1947); Bolliger, Ulrich, ibid. 35, 93 (1952). Stereochemical study: S. Tsukamoto et al., J. Chem. Soc. Perkin Trans. 1 1988, 2621. Review: R. C. Elderfield in W. W. Pigman, M. L. Wolfrom, Advances in Carbohydrate Chemistry vol. I (Academic Press, New York, 1945) pp 159-164.