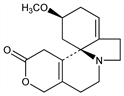
3175. Dihydro-β-erythroidine
Nomenclature
CAS number: 23255-54-1
(3β)-1,6-Didehydro-14,17-dihydro-3-methoxy-16(15H)-oxaerythrinan-15-one; 12,13-didehydro-2,7,13,14-tetrahydro-α-erythroidine. C16H21NO3; mol wt 275.34.
C 69.79%, H 7.69%, N 5.09%, O 17.43%.
Description and references
The more active of the two isomers that constitute the principal alkaloidal fraction of seeds from several Erythrina spp. In contrast to curare, q.v., it retains its ability to block neuromuscular transmission even when administered orally: K. Unna, J. G. Greslin, J. Pharmacol. 80, 53 (1944); K. Unna et al., ibid. 39. Prepd by catalytic hydrogenation of β-erythroidine or a salt of β-erythroidine: Folkers, Koniuszy, US 2370651 (1945 to Merck & Co.). Structure: Boekelheide et al., J. Am. Chem. Soc. 75, 2550 (1953). Configuration: Hanson, Proc. Chem. Soc. London 1963, 52.