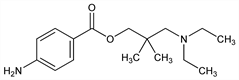
3221. Dimethocaine
Nomenclature
CAS number: 94-15-5
3-(Diethylamino)-2,2-dimethyl-1-propanol p-aminobenzoate; 3-diethylamino-2,2-dimethylpropyl p-aminobenzoate; 1-aminobenzoyl-2,2-dimethyl-3-diethylaminopropanol; p-aminobenzoate of diethylaminoneopentyl alcohol; Larocaine (Roche). C16H26N2O2; mol wt 278.39.
C 69.03%, H 9.41%, N 10.06%, O 11.49%.
Description and references
Prepn: Mannich, Wilder, Ber. 65, 378 (1932); C. Mannich, US 1889678 (1932). Pharmacology: O. Gebner et al., Arch. Exp. Pathol. Pharmakol. 168, 447 (1932); and toxicology review: A. D. Hirschfelder, R. N. Bieter, Physiol. Rev. 12, 190-282 (1932).
Derivative
Hydrochloride.Nomenclature
CAS number: 553-63-9
C16H26N2O2.HCl; mol wt 314.85.
C 61.04%, H 8.64%, N 8.90%, O 10.16%, Cl 11.26%.
Properties
Minute crystals or powder, sometimes fine leaflets. Bitter taste. mp 196-197°. One part dissolves in 3 parts water at 20°, more sol in hot water. One part dissolves also in 10 parts of cold alcohol, in 5 parts of boiling alcohol. Practically insol in ether, oils, fats. Aq solns are slightly acid to litmus, and may be sterilized by heating at 105° for 10 minutes. MLD in mice, rabbits (g/kg): 0.30, 0.15 s.c.; 0.04, 0.015 i.v. (Hirschfelder, Bieter).Therapeutic Category
Anesthetic (local).
Keywords
Anesthetic (Local)