3309. Diphenhydramine
Nomenclature
Description and references
Antihistamine with sedating and antiemetic effects. Prepn: G. Rieveschl, Jr., US 2421714 (1947 to Parke, Davis); see also H. Martin et al., US 2397799 (1946 to Geigy). Toxicology study: O. M. Gruhzit, R. A. Fisken, J. Pharmacol. Exp. Ther. 89, 227 (1947). Comprehensive description of the hydrochloride: I. J. Holcomb, S. A. Fusari, Anal. Profiles Drug Subs. 3, 173-232 (1974). Clinical pharmacokinetics: J. M. Scavone et al., J. Clin. Pharmacol. 38, 603 (1998) PubMed. Clinical trial in insomnia: K. Rickels et al., ibid. 23, 235 (1983). Spectrofluorometric determn in pharmaceutical preparations: I. Pascual Reguera et al., Anal. Sci. 20, 799 (2004) DOI PubMed. CE-MS determn in urine: A. Baldacci et al., Electrophoresis 25, 1607 (2004) DOI PubMed.
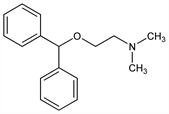
Properties
bp2.0 150-165°.Derivative
Hydrochloride.Nomenclature
Properties
Crystals from abs alcohol + ether, mp 166-170°. Bitter taste. Slowly darkens on exposure to light. Stable under ordinary conditions. One gram dissolves in 1 ml water, 2 ml alcohol, 2 ml chloroform, 50 ml acetone. Very slightly sol in benzene, ether. pH of 1% aq soln about 5.5. The aq soln forms a pink precipitate with satd Reinecke's salt soln. LD50 orally in rats: 500 mg/kg (Gruhzit, Fisken).Derivative
Di(acefyllinate).Nomenclature
Description and references
Prepn: M. Mizier, US 2942000 (1960 to Delagrange).
Properties
Crystals, mp 168-170°. Sol in alc; sparingly sol in water.Therapeutic Category
Antihistaminic; sedative, hypnotic; antiemetic.
Therapeutic Category (Veterinary)
Antihistaminic; antiemetic.Keywords
Antihistaminic; Aminoalkylethers; Sedative/Hypnotic; Antiemetic