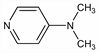
3389. DMAP
Nomenclature
Description and references
Basic nucleophilic catalyst. Prepn: E. Koenigs et al., Ber. 58, 2571 (1925). Fluorescence spectra: S. Mishina et al., J. Photochem. Photobiol. A 141, 153 (2001). Use in Baylis-Hillman chemistry: K. Y. Lee et al., Bull. Korean Chem. Soc. 23, 659 (2002); in olefin hydroxylations: Q. Yao, Org. Lett. 4, 2197 (2002); in regioselective acylations: T. Kurahashi et al., Tetrahedron 58, 8669 (2002). Review of early literature and use in acylation chemistry: G. H"ofle et al., Angew. Chem. Int. Ed. 17, 569-583 (1978); U. Ragnarsson, L. Grehn, Acc. Chem. Res. 31, 494-501 (1998); and of alkylation: E. F. V. Scriven, Chem. Soc. Rev. 12, 129-161 (1983). Brief description and synthetic uses: A. Hassner, "4-(Dimethylamino)pyridine" in Encyclopedia of Reagents for Organic Synthesis 3, L. A. Paquette, Ed. (John Wiley & Sons, New York, 1995) pp 2022-2023.