3440. Doxycycline
Nomenclature
Description and references
Prepn of family of 6-deoxytetracyclines: C. R. Stephens et al., J. Am. Chem. Soc. 80, 5324 (1958). See also: McCormick, Jensen, US 3019260 (1962 to Am. Cyanamid). Prepn, separation and configuration of 6α- and 6β-epimers: M. S. von Wittenau et al., J. Am. Chem. Soc. 84, 2645 (1962); C. R. Stephens et al., ibid. 85, 2643 (1963). Prepn of 6α-deoxyoxytetracycline: R. K. Blackwood et al., US 3200149 (1965 to Pfizer). 1H-NMR study: M. S. von Wittenau, R. K. Blackwood, J. Org. Chem. 31, 613 (1966). Biological properties: English, Proc. Soc. Exp. Biol. Med. 122, 1107 (1966). Pharmacology: Fabre, Chemotherapia 11, 73 (1966); Gibaldi, ibid. 12, 265 (1967). Toxicity of hyclate: Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Clinical trial in prophylaxis of leptospirosis: E. T. Takafuji et al., N. Engl. J. Med. 310, 497 (1984). Clinical trial in periodontitis: A. M. Polson et al., J. Periodontol. 68, 110, 119 (1997). Review: C. Edwards in Pharmacological and Biochemical Properties of Drug Substances vol. 2, M. E. Goldberg, Ed. (Am. Pharm. Assoc., Washington, DC, 1979) pp 305-332.
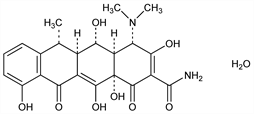
Derivative
Hydrochloride hemiethanolate hemihydrate.Nomenclature
Properties
Light yellow, crystalline powder from ethanol + HCl. Chars without melting at about 201°. [α]D25 -110° (c = 1 in 0.01N methanolic HCl). uv max (0.01N methanolic HCl): 267, 351 nm (log ε 4.24, 4.12). Sol in water. The alcohol and water of crystallization are lost by drying at 100° under reduced pressure. More active biologically than the corresponding 6β-epimer hydrochloride (Wittenau, 1962). LD50 i.p. in rats: 262 mg/kg (Goldenthal).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Tetracyclines