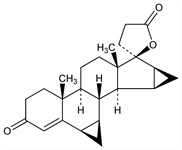
3454. Drospirenone
Nomenclature
Description and references
Synthetic progestogen exhibiting antimineralocorticoid and antiandrogenic activity. Prepn: R. Wiechert et al., DE 2652761; eidem, US 4129564 (both 1978 to Schering AG); D. Bittler et al., Angew. Chem. 94, 718 (1982). HPLC determn in human plasma: W. Krause, U. Jakobs, J. Chromatogr. 230, 37 (1982). Pharmacological profile: P. Muhn et al., Contraception 51, 99 (1995). Review of synthesis: H. Laurent et al., J. Steroid Biochem. 19, 771-776 (1983); of pharmacology and clinical experience: W. Oelkers, Mol. Cell. Endocrinol. 217, 255-261 (2004).
Properties
mp 201.3°. [α]D22 -182° (c = 0.5 in chloroform). uv (methanol): 265 nm (ε 19000).Derivative
Mixture with ethinyl estradiol.Nomenclature
Angeliq (Schering AG); Yasmin (Schering AG); Yaz (Berlex).Description and references
Clinical trial as oral contraceptive: K. S. Parsey, A. Pong, Contraception 61, 105 (2000); in treatment of menopausal symptoms: R. Schürmann et al., Climacteric 7, 189 (2004).
Therapeutic Category
Progestogen. In combination with estrogen as oral contaceptive and in treatment of menopausal symptoms.
Keywords
Progestogen; Contraceptive (Oral)