3457. Droxidopa
Nomenclature
Description and references
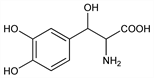
Synthetic amino acid precursor of norepinephrine, q.v. Prepn of racemate: K. W. Rosenmund, H. Dornsaft, Ber. 52B, 1734 (1919). Separation and resolution of diastereomers: B. Hegedüs et al., Helv. Chim. Acta 58, 147 (1975); B. Hegedüs, A. Krasso, US 3920728 (1975 to Hoffmann-La Roche). Improved process for production: N. Ohashi et al., US 4319040 (1982 to Sumitomo). Pharmacology of stereoisomers: G. Bartholini et al., J. Pharmacol. Exp. Ther. 193, 523 (1975). Clinical pharmacology of l-threo-form and clinical evaluation in familial amyloid polyneuropathy (FAP): T. Suzuki et al., Eur. J. Clin. Pharmacol. 17, 429 (1980). Reversed-phase chromatography determn in plasma and urine: F. Boomsma et al., J. Chromatogr. 427, 219 (1988). Pharmacokinetics in FAP: T. Suzuki et al., Eur. J. Clin. Pharmacol. 23, 463 (1982); in parkinsonism: T. Suzuki et al., Neurology 34, 1446 (1984). Metabolism to norepinephrine: T. Suzuki et al., Life Sci. 36, 435 (1985). Clinical studies in Parkinson's disease: N. Ogawa et al., J. Med. 16, 525 (1985); H. Narabayashi et al., Adv. Neurol. 45, 593 (1986).
Properties
Crystals from ethanol and ether, mp 232-235° (dec). [α]D20 -39° (c = 1 in 1N aq HCl). Also cited as crystals from water and l-ascorbic acid, mp 229-232° (dec) (Ohashi). [α]D20 -42.0° (c = 1 in 1N aq HCl).Therapeutic Category
Antiparkinsonian.
Keywords
Antiparkinsonian