3466. DuPHOS
Description and references
Series of chiral bis(phospholane) compounds with C2 symmetry used as rhodium complexes for catalysis. Prepn: M. J. Burk, J. Am. Chem. Soc. 113, 8518 (1991); and configuration: idem et al., ibid. 115, 10125 (1993). Use in asymmetric synthesis of warfarin: A. Robinson et al, Tetrahedron Lett. 37, 8321 (1996); hydrogena-tions of β-(acylamino)acrylates: G. Zhu et al., J. Org. Chem. 64, 6907 (1999).
Derivative
(R,R)-Me-DuPHOS.Nomenclature
CAS number: 147253-67-6
[2R-[1(2′R*,5′R*),2α,5β]]-1,1′-(1,2-Phenylene)bis[2,5-dimethylphospholane]. C18H28P2; mol wt 306.36.
C 70.57%, H 9.21%, P 20.22%.
Derivative
(S,S)-Me-DuPHOS.Nomenclature
CAS number: 136735-95-0
(S,S)-1,2-Bis(2,5-dimethylphospholano)benzene. C18H28P2; mol wt 306.36.
C 70.57%, H 9.21%, P 20.22%.
Properties
Colorless crystals from methanol, mp 79-81°. [α]D25 +476± 5° (c = 1 in hexane).Derivative
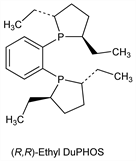
(R,R)-Et-DuPHOS.Nomenclature
CAS number: 136705-64-1
(2R,2′R,5R,5′R)-1,1′-(1,2-Phenylene)bis[2,5-diethylphospholane]. C22H36P2; mol wt 362.47.
C 72.90%, H 10.01%, P 17.09%.