3493. Ecgonine
Nomenclature
CAS number: 481-37-8
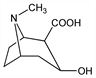
[1R-(exo,exo)]-3-Hydroxy-8-methyl-8-azabicyclo[3.2.1]octane-2-carboxylic
acid; 3β-hydroxy-1αH,5αH-tropane-2β-carboxylic
acid. C9H15NO3; mol wt 185.22.
C 58.36%, H 8.16%, N 7.56%, O 25.91%.
Description and references
The principal part of the cocaine molecule; naturally occurring as the l-form. Obtained by hydrolysis of cocaine: Willst"atter et al., Ann. 434, 111 (1923); Bell, Archer, J. Am. Chem. Soc. 82, 4642 (1960). From coca leaves: de Jong, Rec. Trav. Chim. 59, 687 (1940). Structure: Gadamer, John, Arch. Pharm. 259, 227 (1921). Stereochemistry: Fodor, Nature 170, 278 (1952); Fodor, Kovács, J. Chem. Soc. 1953, 724. Synthesis: Willst"atter, Bommer, Ann. 422, 15 (1920). Review: Stoll, Jucker, Angew. Chem. 66, 376 (1954).
Derivative
l-Form monohydrate.Nomenclature
CAS number: 5796-30-5
Properties
Triboluminescent, monoclinic prisms from alc, mp 198° (anhydr, dec 205°). [α]D15 -45° (c = 5). Neutral to litmus. pKa 11.11; pKb 11.22. One gram dissolves in 5 ml water, 67 ml alc, 20 ml methanol, 75 ml ethyl acetate. Sparingly sol in acetone, ether, benzene, chloroform, petr ether.Derivative
Hydrochloride.Nomenclature
CAS number: 5796-31-6
C9H15NO3.HCl; mol wt 221.68.
C 48.76%, H 7.27%, N 6.32%, O 21.65%, Cl 15.99%.
Properties
Triclinic plates from water, mp 246°. [α]D15 -59° (c = 10). Sol in water; slightly in alc.Derivative
dl-Form trihydrate.Properties
Plates from 90% alcohol, mp 93-118° (anhydr dec 212°).Note
This is a controlled substance: 21 CFR, 1308.12.Therapeutic Category
Anesthetic (topical).
Keywords
Anesthetic (Local)