3495. Echinenone
Nomenclature
CAS number: 432-68-8
β,β-Caroten-4-one; 4-oxo-β-carotene; 4-keto-β-carotene; aphanin; myoxanthin. C40H54O; mol wt 550.86.
C 87.21%, H 9.88%, O 2.90%.
Description and references
Carotenoid pigment occurring in algae. Isoln from Aphanizomenon flos-aqua: Tischer, Z. Physiol. Chem. 251, 109 (1938); 260, 257 (1939); from Paracentrotus lividus: Lederer, Compt. Rend. 201, 300 (1935); from Oscillatoria rubrescens: Heilbron, Lythgoe, J. Chem. Soc. 1936, 1376. Structure: Goodwin, Taha, Biochem. J. 47, 244 (1950); 48, 513 (1951); Goodwin, ibid. 63, 481 (1956); Ganguly et al., Arch. Biochem. Biophys. 60, 345 (1956). Synthesis: Akhtar, Weedon, J. Chem. Soc. 1958, 3986; 1959, 4058.
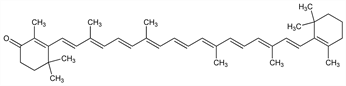
Properties
Orange-red crystals from benzene + methanol, mp 178-180°. Absorption max (chloroform): 472-478 nm. Freely sol in carbon disulfide, chloroform, benzene. Slightly sol in pyridine, ether. Practically insol in methanol. Provitamin A activity 54% of that of all-trans-β-carotene.Derivative
Oxime.C40H55NO; mol wt 565.87.
C 84.90%, H 9.80%, N 2.48%, O 2.83%.