3498. Echinopsine
Nomenclature
CAS number: 83-54-5
1-Methyl-4(1H)-quinolinone; 1-methyl-4(1H)-quinolone; 1,4-dihydro-1-methyl-4-oxoquinoline; N-methyl-4-quinolone. C10H9NO; mol wt 159.18.
C 75.45%, H 5.70%, N 8.80%, O 10.05%.
Description and references
Isoln from Echinops ritro L. and other spp of Echinops, Compositae: Greshoff, Rec. Trav. Chim. 19, 360 (1900); Ban'kovskii et al., Dokl. Akad. Nauk SSSR 148, 1073 (1963). Structure: Sp"ath, Kolbe, Monatsh. Chem. 43, 469 (1923). Synthesis: Kondo, Ikawa, J. Pharm. Soc. Jpn. 51, 702 (1931); Allison et al., J. Chem. Soc. 1954, 403; King, Abramo, J. Org. Chem. 23, 1609 (1958); Kamiya, Chem. Pharm. Bull. 10, 669 (1962). Simple synthesis: J. R. Merchant, V. Shankaranarayan, Chem. Ind. (London) 1979, 320. Pharmacology: A. D. Turova et al., Pharmacol. Toxicol. 20, 236 (1957).
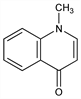
Properties
Needles from benzene, mp 152°. One gram dissolves in about 60 ml water, 6 ml boiling water. Soluble in alcohol, chloroform, hot benzene; slightly sol in ether. LD100 s.c. in mice: 600 mg/kg (Turova).Derivative
Hydrochloride.C10H9NO.HCl; mol wt 195.65.
C 61.39%, H 5.15%, N 7.16%, O 8.18%, Cl 18.12%.