3516. Edrophonium Chloride
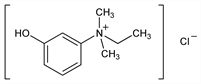
Nomenclature
Description and references
Cholinesterase inhibitor. Prepn: J. A. Aeschlimann, A. Stempel, US 2647924 (1953 to Hoffmann-La Roche). LC determn in biological fluids: M. G. M. DeRuyter et al., J. Chromatogr. 183, 193 (1980). Clinical evaluation for reversal of neuromuscular blockade during anesthesia: D. M. Fisher et al., Anesthesiology 61, 428 (1984); T. Suzuki et al., Can. J. Anaesth. 50, 879 (2003). Clinical study in supraventricular tachycardia: J. D. Cantwell et al., Arch. Intern. Med. 130, 221 (1972). Review of diagnostic use in myasthenia gravis: R. M. Pascuzzi, Semin. Neurol. 23, 83-88 (2003).
Properties
Crystals from isopropanol, dec 162-163°. pH of 1% aq soln 4-5. Very sol in water; freely sol in alcohol. Insol in chloroform, ether.Derivative
Edrophonium Bromide.Nomenclature
Properties
Crystals from ethanol + ether, dec 151-152°. Bitter taste. Sol in water (more than 10%). Moderately sol in alcohol. Practically insol in ether. Solns are stable.Therapeutic Category
Cholinergic; reversal agent for neuromuscular blockade; antidote to curare; diagnostic aid (myasthenia gravis).
Therapeutic Category (Veterinary)
Cholinergic; reversal agent for neuromuscular blockade; diagnostic aid (myasthenia gravis).Keywords
Antidote (Curare); Cholinesterase Inhibitor; Cholinergic; Diagnostic Aid; Neuromuscular Block Reversant