3531. Eicosapentaenoic Acid
Nomenclature
Description and references
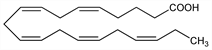
Important polyunsaturated fatty acid of the marine food chain that serves as a precursor for the prostaglandin-3 and thromboxane-3 families. It differs from arachidonic acid, q.v. (the eicosatetraenoic acid that is a precursor for the prostaglandin and thromboxane-2 families) by the extra double bond between the third and fourth carbons from the “methyl end” of the molecule. Isoln from cod liver oil: E. Klenk, D. Eberhagen, Z. Physiol. Chem. 307, 42 (1957). Enzymatic conversion to prostaglandin E3: S. Bergstr"om et al., J. Biol. Chem. 239, PC 4006 (1964). Effects on role of platelets in thrombosis: K. C. Srivastava et al., Biochem. Exp. Biol. 16, 317 (1980). Effects on prostacyclin-like material in human umbilical vasculature: J. Dyerberg, K. A. Jorgensen, Artery 8, 12 (1980). A possible relationship between diets rich in EPA in marine oils and low rates of ischemic heart disease has been proposed: H. O. Bang et al., Am. J. Clin. Nutr. 33, 2657 (1980); J. Dyerberg, Philos. Trans. R. Soc. London Ser. B 294, 373 (1981). Clinical evaluation of lipid lowering effect: Y. Nagakawa et al., Atherosclerosis 47, 71 (1983); of use in rheumatoid arthritis: J. M. Kremer et al., Ann. Intern. Med. 106, 497 (1987); of use in Raynaud's phenomenon: R. A. DiGiacomo et al., Am. J. Med. 86, 158 (1989).
Properties
Colorless oil. nD20 1.49865.Derivative
Ethyl ester.Nomenclature
Therapeutic Category
Antilipemic.
Keywords
Antilipemic